What is Carnot Cycle?
The Carnot cycle was first proposed by a French engineer in 1824 and was expanded upon by others in 1830s and 1840s. It is an ideal cycle in which, the working medium receives the heat energy from the high temperatures and rejects the heat at the lower temperature. This cycle laid the foundation for the second law of thermodynamics and introduced the concept of reversibility.
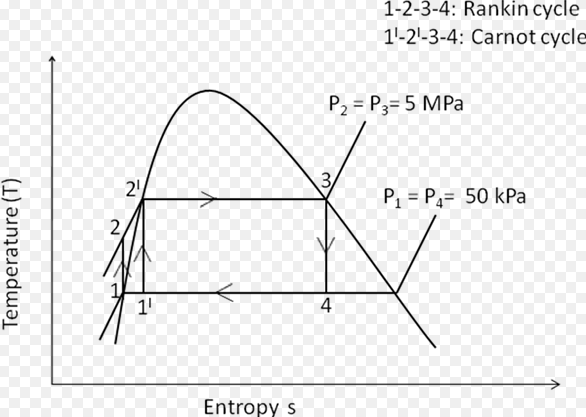
Carnot cycle is an excellent yardstick to compare various thermodynamic cycles on theoretical basis. Nevertheless, all practical cycles, differ significantly from Carnot cycle.
The Carnot cycle depends on the temperature of the heat source and heat sink only and is independent of the type of working fluid. The higher the temperature of the heat source the lower the temperature of the heat sink, the more the efficiency.
The Carnot cycle consist of four major processes, which include:
- A reversible isothermal gas expansion process. In this process the engine is connected to the thermal source and that the expanding gas absorbs heat from the source and does work on the surroundings. The temperature of the gas remains constant.
- A reversible adiabatic gas expansion process. The system is adiabatic, meaning no heat transfer is possible. In this process, the system is thermally insulated and the gas continues to expand and do work on surroundings, which causes the system to cool to a lower temperature.
- A reversible isothermal gas compression process. In this process, surroundings do work to the gas at lower temperature and causes a loss of heat.
- A reversible adiabatic gas compression process. In this process, the system is thermally insulated. Surroundings continue to do work to the gas, which causes the temperature to rise back to higher temperature.
What is Rankine Cycle?
Rankine cycle was described in 1859 by the Scottish enigineer William J.M Rankine. The cycle closely describes the process by which steam-operated heat engines commonly found in thermal power generation plants generate power.
Power depends on the temperature difference between a heat source and a cold source. The higher the difference, the more mechanical power can be efficiently extracted out of heat energy.
The efficiency of the Rankine cycle is limited by the high heat of vaporization of the working fluid. Also, unless the pressure and temperature reach super critical levels in the steam boiler, the temperature range the cycle can operate over is quite small.
In the Rankine cycle, the working substance of the engine undergoes four successive changes that is; heating at constant pressure, converting the liquid to vapour; reversible adiabatic expansion, performing work (as by the driving turbine), cooling at constant pressure, condensing the vapour to liquid and reversible adiabatic compression, pumping the liquid back to the boiler. The processes can be explained as below:
Also Read: Difference Between 2 Stroke Engine And 4 Stroke Engine
Difference Between Carnot And Rankine Cycle In Tabular Form
| BASIS OF COMPARISON | CARNOT | RANKINE CYCLE |
| Description | Carnot cycle is a theoretical cycle. Its efficiency at its highest between two temperature difference. | Rankine cycle is a practical cycle of turbine and steam engine. |
| Working Substance | Carnot cycle uses air as the working substance. | Rankine cycle uses water as the working substance. |
| Heat Addition | In Carnot cycle, heat addition is done by keeping the temperature constant. | In Rankine cycle, heat addition is done by keeping pressure constant. |
| Heat Rejection | In Carnot cycle, heat rejection is done by keeping temperature constant. | In Rankine cycle, heat rejection is done by keeping pressure constant. |
| Ideal for | Carnot cycle is an ideal cycle for heat engine. | Rankine cycle is an ideal cycle for vapour power cycle. |
| Efficiency | Carnot cycle has the highest efficiency between two temperature differences. | Rankine has a lower efficiency when compared to Carnot cycle, though in real life, Carnot cycle has many advantages than Rankine cycle. |
| Pressure of the Working Fluid | In Carnot cycle, the pressure of working fluid is raised from the condenser pressure to the boiler pressure. | In Rankine cycle, saturated liquid is pumped to the boiler with a pump. |
Also Read: Difference Between Diesel Cycle And Otto Cycle